In this post
The creation of electrical charge is a fundamental part of electricity. Most electrical charge is produced due to the movement of negatively charged particles.
Charges within an atom
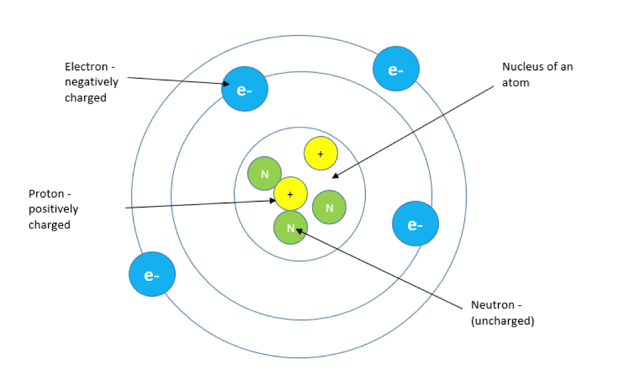
Every atom contains sub-atomic particles known as protons, neutrons and electrons. These sub-atomic particles have different charges and masses. Protons are positively charged particles, found in the nucleus of an atom. Neutrons are also found in the atom’s nucleus but they are neutral which means that they have no charge.
Electrons are negatively charged particles, found orbiting the nucleus of the atom in energy shells.
| Particle | Position in the atom | Charge |
| Proton | Nucleus | +1 |
| Neutron | Nucleus | 0 |
| Electron | Energy shells | -1 |
The number of protons present in the nucleus of an atom is equal to the number of electrons that orbit the nucleus. This means that the atom has no overall charge because there are as many positively charged particles as there are negatively charged particles. Atoms are therefore neutral.
Since the electrons orbit the nucleus in shells which increase in distance away from the nucleus, the outermost electrons are the most likely to be lost or gained. If an atom gains electrons it becomes negatively charged and if it loses electrons it becomes positively charged.
An atom which has lost or gained electrons to become charged is known as an ion. The number of electrons lost or gained determines the overall charge on the ion.
It is the loss of electrons by an atom which creates electricity. Electrical current is the flow of negatively charged electrons through a circuit. Electrons are removed from an atom and are passed along or through a conductor (such as a metal wire) towards an area of positive charge.

Electric currents through conductors and insulators
Current is a measurement of flow of charge which is carried through a circuit by negatively charged electrons. The ease at which the flow passes through an object depends on if the object is a good conductor or not. A good conductor of electricity is a type of material which allows negatively charged electrons to move freely through it.
In conductors, some electrons are free to drift between the atoms. Under normal circumstances, this drifting is considered random. This is because the number of electrons flowing in any one direction is roughly equal to the number of electrons flowing in the opposite direction. Therefore, there is no overall flow of charge. However, if a cell or battery is connected across the conductor, more of the electrons now flow in the direction away from the negative terminal and towards the positive terminal. This is known as a net flow of charge.
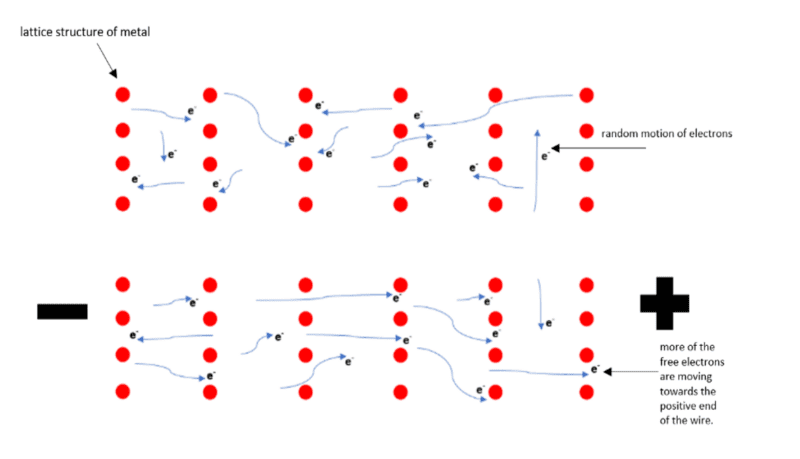
Typically, electrons flow through metals easier than other materials and would not flow well at all through plastics. Metals such as copper, gold, silver and aluminium are good electrical conductors.
An insulator is a type of material which does not conduct electricity. In insulators, all the electrons are held tightly in position and are unable to move from atom to atom. Charges are therefore unable to pass through insulators due to the electrons not being able to move freely.
As plastics do not conduct electricity, they are good electrical insulators. This is why plastic is used to coat the wiring in electrical supplies so as to prevent electrocution. Other examples of good electrical insulators are glass, rubber and wood.



